
Coronavirus disease (COVID-19)
- 3 апреля 2020
- Article 83(3) of Regulation (EC) No 726/2004, azithromycin, chloroquine, clinical data, clinical trials, common logo, compassionate use, coronavirus, COVID-19, CT, disease, efficacy, falsified, guidelines, heart rhythm problems, hydroxychloroquine, IMP, interferon beta, investigational, lopinavir, medicines, MERS-CoV, monoclonal antibodies, nonclinical data, online, pandemic, pharmacies, prevent, remdesivir, ritonavir, safety, SARS-CoV, SARS-Cov-2, serious side effects, treat, treatments, vaccine, Veklury, WHO, безопасность, вакцина, гидроксихлорохин, интернет-аптека, интерферон бета, исследуемое лекарственное средство, клинические исследования, коронавирусная инфекция, лечение, лопинавир, моноклональные антитела, общий логотип, онлайн аптека, пандемия, профилактика, ремдесивир, ритонавир, терапия, фальсифицированные лекарственные препараты, хлорохин, эффективность,
Treatments and vaccines against
COVID-19
Update [03/07/2020]
Remdesivir (Veklury) is the first authorised medicine in the EU
to treat COVID-19!
Update [02/07/2020]
The first COVID-19 treatment has been recommended for EU authorization!
EMA’s human medicines committee (CHMP) has recommended granting
a conditional marketing authorisation to Veklury (remdesivir).
EMA is urging the general public not to buy medicines from unauthorised websites and other vendors aiming to exploit fears and concerns during the ongoing pandemic of coronavirus disease (COVID-19).
Vendors may claim that their products can treat or prevent COVID-19 or may appear to provide easy access to legitimate medicines that are otherwise not readily available. Such products are likely to be falsified medicines.
Keeping safe when buying medicines
- Falsified medicines can cause serious harm.
- When buying over the internet, only use registered online pharmacies.
- Check that the online pharmacy you are using has the common logo.
- Click on the logo and confirm that the online pharmacy is listed on the national authority website.
- Do not buy medicines advertised as cures or preventive treatments for COVID-19. To treat COVID-related symptoms such as fever, discuss with your doctor or follow advice from authorities.
Supporting the rapid development and approval of effective and safe treatments for and vaccines against COVID-19 is EMA’s top priority to help save lives during the pandemic. Over recent weeks and months, the Agency has engaged with many developers of therapeutic medicines and there are a number of developments underway. However, at this point, on the basis of the preliminary data presented to the Agency, no medicine has yet demonstrated efficacy in treating COVID-19.
Among the potential treatments for COVID-19 that are undergoing clinical trials to assess their safety and efficacy against the disease are:
- remdesivir (authorised as treatment against COVID-19);

Product information (PI) on Veklury (remdesivir) is available here.
NEW! Update [08/12/2020]
EMA is aware that the World Health Organization (WHO) has updated its guidelines advising against the use of remdesivir in hospitalised patients with COVID-19, regardless of disease severity based on a recent meta-analysis.
According to WHO, the certainty for the results of the meta-analysis is low and the evidence did not prove that remdesivir has no benefit. In reaching its recommendations, the WHO also considered implications on health resources in view of remdesivir’s cost and the need for it to be given intravenously. The WHO recognised that more research is needed, especially in certain groups of patients, and supported continued enrolment in trials evaluating remdesivir.
EMA has requested the full Solidarity data from WHO and the marketing authorisation holder. Once the data are available, EMA will assess the evidence, together with other relevant data, to see if any changes are needed to the marketing authorisation of Veklury (remdesivir) in the EU.
Update [03/07/2020]
On 3 July 2020, the European Commission granted a conditional marketing authorisation for remdesivir (Veklury), for the treatment of COVID-19 in adults and adolescents from 12 years of age with pneumonia who require supplemental oxygen.
This follows the recommendation of the CHMP from 25 June 2020 and allows the medicine to be marketed in the EU.
Data on remdesivir were assessed in an exceptionally short timeframe through a rolling review procedure, an approach used by EMA during public health emergencies to assess data as they become available. This has enabled the authorisation to be granted quickly in the context of the coronavirus crisis, within one week after EMA’s recommendation, compared to the usual 67 days.
From 30 April 2020, the CHMP began assessing data on quality and manufacturing, nonclinical data, preliminary clinical data and supporting safety data from compassionate use programmes, well in advance of the submission of the marketing authorisation application on 5 June.
The assessment of the dossier has concluded with recommendation of the CHMP from 25 June 2020 on granting a conditional marketing authorisation, which is mainly based on data from study NIAID-ACTT-1, sponsored by the US National Institute of Allergy and Infectious Diseases (NIAID), plus supporting data from other studies on remdesivir.
Taking into consideration the available data, the Agency considered that the balance of benefits and risks had been shown to be positive in patients with pneumonia requiring supplemental oxygen; i.e., the patients with severe disease. Remdesivir is given by infusion (drip) into a vein and its use is limited to healthcare facilities in which patients can be monitored closely; liver and kidney function should be monitored before and during treatment, as appropriate. Treatment should start with a 200-mg infusion on the first day, followed by one 100-mg infusion a day for at least 4 days and no more than 9 days.
Remdesivir is now authorised for a conditional marketing authorisation, one of the EU regulatory mechanisms created to facilitate early access to medicines that fulfil an unmet medical need, including those for emergency situations in response to public health threats such as the current pandemic.
THE END
During an extraordinary virtual meeting held on 2 April 2020, EMA’s human medicines committee (CHMP) gave recommendations on how the investigational antiviral medicine remdesivir should be used for treating coronavirus disease (COVID-19) in compassionate use programmes in the European Union.
Compassionate use programmes, which are set up at the level of individual EU Member States, are intended to give patients with a life-threatening, long-lasting or seriously disabling disease and no available treatment options, access to treatments that are still under development and that have not yet received a marketing authorisation.
In this case Estonia, Greece, the Netherlands and Romania on 25 and 26 March 2020 requested an opinion from the CHMP on the conditions under which early access to remdesivir through compassionate use in accordance with Article 83(3) of Regulation (EC) No 726/2004 could be given to patients with COVID-19. In severe cases, COVID-19 can cause pneumonia, severe acute respiratory syndrome, multi-organ failure and death.
The Applicant is proposing the use of Remdesivir (RDV), when used as part of a compassionate use programme, for the treatment of patients with COVID-19 who require invasive mechanical ventilation. Such patients have been reported to have a mortality at and above 50%. There are no specific therapies with established efficacy and safety for the treatment of COVID-19. Thus, the target condition fulfils criteria for the compassionate use insofar as there is life-threatening illness which cannot be treated satisfactorily with any currently authorised medicine.

Empirical nonclinical data on antiviral activity of remdesivir on SARS-CoV-2 is currently limited to a few in vitro observations and is mainly extrapolated from in vitro and in vivo studies with other coronavirus types (i.e. SARS-CoV and MERS-CoV), that are presumed to have similar pathogenesis and viral susceptibility as does COVID-19 and SARS-CoV-2. These studies indicate that prophylactic treatment is more effective than when RDV is given after viral challenge. Furthermore, there are no data on the initiation of treatment more than one day after challenge.
There are presently no clinical efficacy data to support the use of remdesivir (RDV) coming from patients with infection for COVID-19. There are currently several ongoing and planned clinical studies with this regard. Furthermore, the safety profile of RDV is incompletely characterised. Nevertheless, the safety profile appears supportive of further studies in COVID - 19. Hepatotoxicity is a clinically identified risk and should be monitored.
Remdesivir is currently not approved for marketing in any country. Consequently, patients with COVID-19 should presently preferably access RDV through clinical trials. However, given the dire prognosis in patients with COVID-19 that require mechanical ventilation, compassionate use of RDV is considered reasonable in such patients that are not able to access a clinical trial.
In the context of the compassionate use of RDV for the above-mentioned targeted population and according to the conditions adopted by the CHMP, it is considered that the benefits outweigh the risks.
- lopinavir/ritonavir (currently authorised as an anti-HIV medicine);
- chloroquine and hydroxychloroquine (currently authorised at national level as treatments against malaria and certain autoimmune diseases such as rheumatoid arthritis);
Chloroquine and hydroxychloroquine are being investigated worldwide for their potential to treat coronavirus disease (COVID-19). However, efficacy in treating COVID-19 is yet to be shown in studies.
It is very important that patients and healthcare professionals only use chloroquine and hydroxychloroquine for their authorised uses or as part of clinical trials or national emergency use programmes for the treatment of COVID-19.
Both chloroquine and hydroxychloroquine can have serious side effects, especially at high doses or when combined with other medicines. They must not be used without a prescription and without supervision by a doctor.
Considering the urgency and the pressure healthcare systems face to save lives during the COVID-19 pandemic, some countries, including the USA and France, have put strict protocols in place to allow the experimental use of these two medicines, for example, in patients with severe forms of COVID-19.
Update [05/06/2020]
On 29 May 2020, the European Medicines Agency (EMA) has published the second reminder of risks of chloroquine and hydroxychloroquine.
EMA is reminding healthcare professionals to closely monitor patients with COVID-19 who are receiving chloroquine or hydroxychloroquine, given the serious side effects that can result from treatment with these medicines.
Several observational studies have reported that chloroquine and hydroxychloroquine are associated with an increased risk of heart problems, a well-known side effect of such treatments, including cardiac arrhythmias and cardiac arrest.
When prescribing these medicines, healthcare professionals should take into account pre-existing heart conditions, uncorrected potassium or magnesium imbalance, and concomitant use with medicines that prolong the QT interval as these factors may make patients more prone to heart rhythm disorders.
In view of the emerging data, some EU countries have suspended or stopped clinical trials investigating chloroquine and hydroxychloroquine in COVID-19 patients. For some trials, including WHO’s large multinational Solidarity trial, enrolment of patients to trial arms with these medicines has been suspended. A preliminary review of the Recovery trial, a large ongoing study on COVID-19 patients, did not identify reasons to suspend or stop the trial.
EMA reiterates that while further analyses of available data are being carried out, chloroquine and hydroxychloroquine should only be used in clinical trials for treatment or prophylaxis of COVID-19 or in national emergency use programmes in hospitalised patients under close supervision. It is important that properly designed, randomised clinical trials can be completed, with adjustments as needed, to generate the necessary evidence on benefits and risks of these medicines in COVID-19.
Update [24/04/2020]
On 23 April 2020, the European Medicines Agency (EMA) has published a reminder of risk of serious side effects with chloroquine and hydroxychloroquine.
Chloroquine and hydroxychloroquine are known to potentially cause heart rhythm problems, and these could be exacerbated if treatment is combined with other medicines, such as the antibiotic azithromycin, that have similar effects on the heart.
Recent studies have reported serious, in some cases fatal, heart rhythm problems with chloroquine or hydroxychloroquine, particularly when taken at high doses or in combination with the antibiotic azithromycin.
In addition to side effects affecting the heart, they are known to potentially cause liver and kidney problems, nerve cell damage that can lead to seizures (fits) and low blood sugar (hypoglycaemia).
These medicines are being used in the context of the ongoing pandemic for treating patients with COVID-19 and investigated in clinical trials. However, clinical data are still very limited and inconclusive, and the beneficial effects of these medicines in COVID-19 have not been demonstrated. Results from large, well-designed studies are needed to make any conclusions.
Some clinical trials currently investigating the effectiveness of chloroquine or hydroxychloroquine in treating COVID-19 use higher doses than those recommended for the authorised indications. While serious side effects can occur with recommended doses, higher doses can increase the risk of these side effects, including abnormal electrical activity that affects the heart rhythm (QT-prolongation).
Healthcare professionals are recommended to closely monitor patients with COVID-19 receiving chloroquine or hydroxychloroquine and to take into account pre-existing heart problems that can make patients more prone to heart rhythm issues. They should carefully consider the possibility of side effects, particularly with higher doses, and exercise extra caution when combining treatment with other medicines such as azithromycin that may cause similar side effects on the heart.
Patients and healthcare professionals are reminded to report any suspected side effects to their national regulatory authorities.
- systemic interferons and in particular interferon beta (currently authorised to treat diseases such as multiple sclerosis);
- monoclonal antibodies with activity against components of the immune system.
More information on the use of monoclonal antibodies to treat COVID-19 you can find here.
The Agency has also had discussions with developers of a dozen potential COVID-19 vaccines. Two vaccines have already entered phase I clinical trials, which are the first trials needed and are carried out in healthy volunteers. In general, timelines for the development of medicinal products are difficult to predict. Based on the information currently available and past experience with vaccine development timeframes, EMA estimates that it might take at least one year before a vaccine against COVID-19 is ready for approval and available in sufficient quantities to enable widespread use. Adequate supply of doses to meet the needs of all EU countries has to be proactively forecast.
More information on vaccines against COVID-19 you can find here.
Availability of medicines during the COVID-19 pandemic –
How to avoid shortages?
Factsheet (European Commission) – Guidelines to optimise supply and avoid medicines shortages

Showing solidarity
- Lifting export bans and restrictions
- Avoiding national stockpiling
- Avoiding that misinformation leads to improper use and unnecessary stockpiling

Ensuring supply
- Increasing and reorganising production
- Ensuring manufacturing continues at full capacity
- Implementing regulatory flexibility
- marketing authorisations and regulatory procedures;
- manufacturing and importation of active pharmaceutical ingredients (APIs) and finished products;
- quality variations;
- labelling and packaging requirements with flexibility to facilitate the movement of medicinal products within the EU;
- good manufacturing practice (GMP) and good distribution practice (GDP) inspections in the context of restrictions linked to COVID-19 pandemic;
For instance, the validity of GMP certificates and time-limited manufacturing and import authorisations, as well as the validity of GDP certificates and time-limited wholesale authorisations will be extended until the end of 2021.
- adaptations that can be applied to the work of the QP in the context of restrictions arising from COVID-19 pandemic (remote batch certification, remote audits of the active substance manufacturer, batch release of investigational medicinal products imported from third countries by relying on a variety of documents);

- and submitting individual case safety reports (ICSR) to EudraVigilance.
Additional guidance is available here for qualified persons for pharmacovigilance (QPPV) and marketing authorisation holders on submitting COVID-19-related ICSRs to EudraVigilance.
The Notification for MedDRA users regarding existing and new terms for coronavirus concepts is available here.
- Monitoring available stocks at national level
- Ensuring necessary support to the wholesale sector
- Fully enforcing the green lanes
- Facilitating air freight and other forms of transport
- Ensuring fair distribution of supply
Regulatory requirements in the context of the COVID-19 pandemic
The European Commission, EMA and the European medicines regulatory network have developed a question-and-answer (Q&A) document to provide guidance to stakeholders on adaptations to the regulatory framework to address challenges arising from the COVID-19 pandemic, with a particular focus on crucial medicines for use in COVID-19 patients.
The document explains some regulatory flexibilities that can be applied to help pharmaceutical companies cope with the consequences of the pandemic, while ensuring a high level of quality, safety and efficacy for medicinal products made available to patients in the EU.
This document provides guidance to marketing authorisation holders of medicinal products for human use (MAH) on regulatory expectations and flexibility during the COVID-19 pandemic.
The measures introduced cover different areas of the regulation of medicines such as:
Some of the measures described are reserved for crucial medicines for use in COVID-19 patients.
When in doubt whether a given medicinal product is a crucial medicine for treatment of COVID - 19 patients, the MAH may contact the relevant competent authorities
(EMA should be contacted for centralised marketing authorisations).
The CMDh has agreed additional questions and answers that provide practical information on how to specifically address and apply the provisions determined by the European Commission for MR/DC procedures.
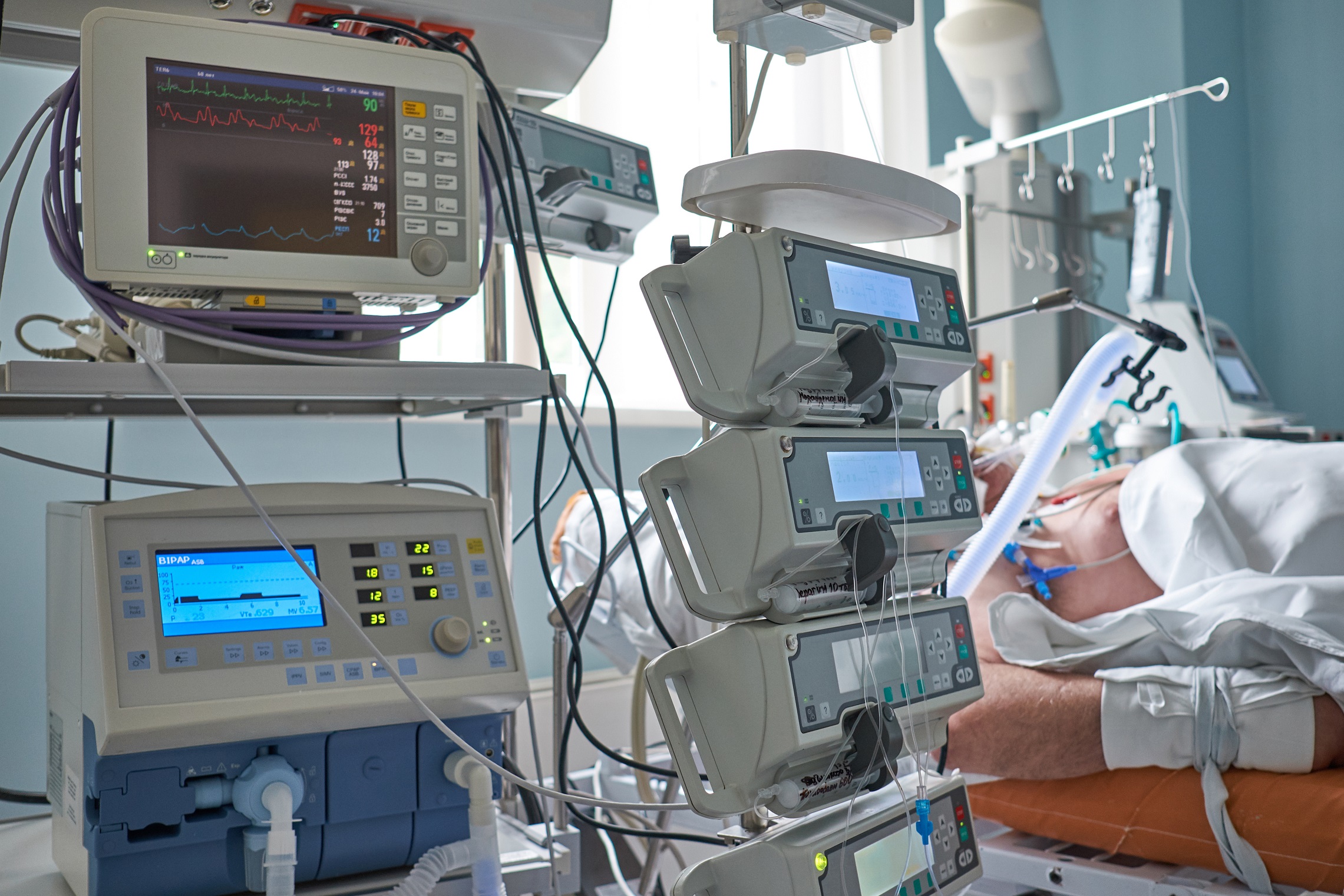
Optimal use of medicines in hospitals
- Equitable distribution of available medicines
- Exchanging hospital protocols to treat patients
- Considering alternative medicines on the basis of hospital protocols and national guidelines
- Extending the expiry dates of medicines
- Considering the use of magistral preparations or veterinary medicines
- Using medicines off label and in clinical trials

Optimisation of sales in community pharmacies to avoid hoarding
- Introducing measures to reassure persons reliant on medication
- Introducing restrictions on sales in community pharmacies
- Limiting online sales of products at risk
- Reassuring patients
COMMUNICATION FROM THE COMMISSION – Guidelines on the optimal and rational supply of medicines to avoid shortages during the COVID-19 outbreak – is available here.
Source: EMA, EC, HMA, ICH





